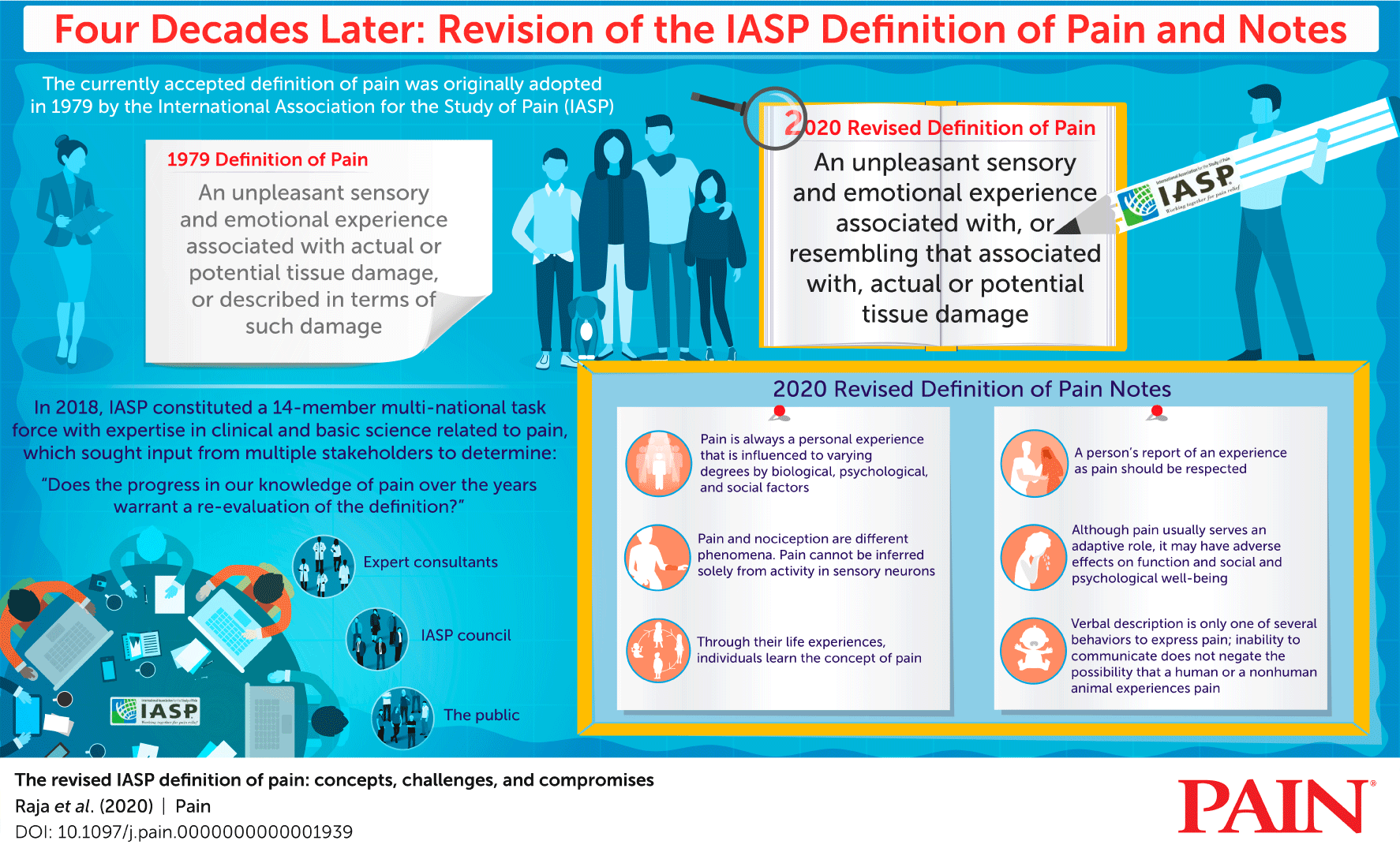
Recently, an updated definition of “pain” was constructed by a multidisciplinary group of leading experts in the field of pain on behalf of the International Association for the Study of Pain, the leading international scientific and professional organization for the field, and published in Pain1, the highest impact journal in the field. In addition to the revised definition, the expert panel proposed a series of notes to accompany it and summarized their findings in a detailed infographic.
The IASP definition of pain and qualifying notes are entirely consistent with the mission and objectives of the Pain Management Collaboratory. Particularly relevant is the acknowledgement that pain is a personal, subjective and multidimensional experience that can be shaped by a range of biological, psychological and social factors. Consistent with this perspective, the Collaboratory’s key objective is to support the conduct of high quality pragmatic clinical trials of nonpharmacological approaches for pain and co-occurring medical and mental health and substance use disorders that are informed by the biopsychosocial model of pain. The pragmatic nature of the trials ensures the broadest feasible representation of veterans and military service members and their dependents with pain who can potentially benefit from these approaches.
A critical feature of the PMC trials is their focus on outcomes important to persons with pain, including reduced pain intensity and improved physical and emotional function. The updated definition and accompanying notes are especially relevant as our pragmatic trials enroll participants based on self-reporting of pain, pain intensity, and pain interference rather than on the presence of a specific diagnosis or biomarker. We recognize the unique life experiences of our trial participants as veterans and military service members and appreciate that an individual’s concept of pain may develop over time and vary by person. We appreciate that an individual may experience challenges of communicating emotion and experience related to pain, particularly through verbal means in face-to-face settings. We are encouraged that a number of our trials are including and/or investigating mechanisms to overcome these potential barriers using technology-driven solutions to support communication of pain experience through asynchronous and non-traditional means.
The successful enactment of the PMC trials in real-world settings enhances the likelihood that interventions demonstrated to be safe and effective in improving these pain-related outcomes can be feasibly integrated into routine clinical care, thus addressing the historical gap between evidence of effectiveness of these approaches and timely and equitable access.
References
- The revised International Association for the Study of Pain definition of pain concepts, challenges, and compromises. Raja, Srinivasa N; Carr, Daniel B; Cohen, Milton; Finnerup, Nanna B; Flor, Herta; Gibson, Stephen; Keefe, Francis J; Mogil, Jeffrey S; Ringkamp, Matthias; Sluka, Kathleen A.; Song, Xue-Jun; Stevens, Bonnie; Sullivan, Mark D.; Tutelman, Perri R; Ushida, Takahirop; Vader, Kyle. doi: 10.1097/j.pain.0000000000001939. Available from: https://journals.lww.com/pain/Abstract/2020/09000/The_revised_International_Association_for_the.6.aspx
